Zinc chloride
 Anhydrous
| |
 Monohydrate
| |

| |
| Names | |
|---|---|
| IUPAC name
Zinc chloride
| |
Other names
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.028.720 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2331 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| ZnCl2 | |
| Molar mass | 136.315 g/mol |
| Appearance | White hygroscopic and very deliquescent crystalline solid |
| Odor | odorless |
| Density | 2.907 g/cm3 |
| Melting point | 290 °C (554 °F; 563 K)[1] |
| Boiling point | 732 °C (1,350 °F; 1,005 K)[1] |
| 432.0 g/100 g (25 °C) 615 g/100 g (100 °C) | |
| Solubility | soluble in ethanol, glycerol and acetone |
| Solubility in ethanol | 430.0 g/100 ml |
| −65.0·10−6 cm3/mol | |
| Structure | |
| Tetrahedral, linear in the gas phase | |
| Pharmacology | |
| B05XA12 (WHO) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Oral toxicity, irritant[2] |
| GHS labelling: | |
  
| |
| Danger | |
| H302, H314, H410 | |
| P273, P280, P301+P330+P331, P305+P351+P338, P308+P310 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
|
LC50 (median concentration)
|
1260 mg/m3 (rat, 30 min) 1180 mg-min/m3[4] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 1 mg/m3 (fume)[3] |
REL (Recommended)
|
TWA 1 mg/m3 ST 2 mg/m3 (fume)[3] |
IDLH (Immediate danger)
|
50 mg/m3 (fume)[3] |
| Safety data sheet (SDS) | External SDS |
| Related compounds | |
Other anions
|
|
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Zinc chloride is an inorganic chemical compound with the formula ZnCl2·nH2O, with n ranging from 0 to 4.5, forming hydrates. Zinc chloride, anhydrous and its hydrates, are colorless or white crystalline solids, and are highly soluble in water. Five hydrates of zinc chloride are known, as well as four forms of anhydrous zinc chloride.[5] All forms of zinc chloride are deliquescent. Zinc chloride finds wide application in textile processing, metallurgical fluxes, and chemical synthesis.[5] In a major monograph, zinc chlorides have been described as "one of the important compounds of zinc."[6]
Structure and properties[edit]
Relative to other metal dihalides, zinc dichloride is unusual in forming several crystalline forms (polymorphs). Four are known: α, β, γ, and δ. Each case features tetrahedral Zn2+ centers.[7]
| Form | Crystal system | Pearson symbol | Space group | No. | a (nm) | b (nm) | c (nm) | Z | Density (g/cm3) |
|---|---|---|---|---|---|---|---|---|---|
| α | tetragonal | tI12 | I42d | 122 | 0.5398 | 0.5398 | 0.64223 | 4 | 3.00 |
| β | tetragonal | tP6 | P42/nmc | 137 | 0.3696 | 0.3696 | 1.071 | 2 | 3.09 |
| γ | monoclinic | mP36 | P21/c | 14 | 0.654 | 1.131 | 1.23328 | 12 | 2.98 |
| δ | orthorhombic | oP12 | Pna21 | 33 | 0.6125 | 0.6443 | 0.7693 | 4 | 2.98 |
Here a, b, and c are lattice constants, Z is the number of structure units per unit cell, and ρ is the density calculated from the structure parameters.[8][9][10]
The orthorhombic form (δ) rapidly changes to one of the other forms on exposure to the atmosphere. A possible explanation is that the OH− ions originating from the absorbed water facilitate the rearrangement.[7] Rapid cooling of molten ZnCl2 gives a glass.[11]
Molten ZnCl2 has a high viscosity at its melting point and a comparatively low electrical conductivity, which increases markedly with temperature.[12][13] As indicated by a Raman scattering study, the viscosity is explained by the presence of polymers,[14]. Neutron scattering study indicated the presence of tetrahedral ZnCl4 centers, which requires aggregation of ZnCl2 monomers as well.[15]
Hydrates[edit]
Various hydrates of zinc chloride are known: ZnCl2(H2O)n with n = 1, 1.33, 2.5, 3, and 4.5.[16] The 1.33-hydrate, previously thought to be the hemitrihydrate, consists of trans-Zn(H2O)4Cl2 centers with the chlorine atoms connected to repeating ZnCl4 chains. The hemipentahydrate, structurally formulated [Zn(H2O)5][ZnCl4], consists of Zn(H2O)5Cl octahedrons where the chlorine atom is part of a [ZnCl4]2- tetrahedera. The trihydrate consists of distinct hexaaquozinc(II) cations and tetrachlorozincate anions; formulated [Zn(H2O)6][ZnCl4]. Finally, the heminonahydrate, structurally formulated [Zn(H2O)6][ZnCl4]·3H2O also consists of distinct hexaaquozinc(II) cations and tetrachlorozincate anions like the trihydrate but has three extra water molecules.[17][18]
Preparation and purification[edit]
Historically, anhydrous zinc chloride has been prepared by treating a suspension of powdered zinc in diethyl ether with hydrogen chloride, followed by drying[19] The overall method remains useful in industry, but without the solvent:[5]
- Zn + 2 HCl → ZnCl2 + H2
Aqueous solutions may be readily prepared similarly by treating Zn metal, zinc carbonate, zinc oxide, and zinc sulfide with hydrochloric acid:[20]
- ZnS + 2 HCl + 4 H2O → ZnCl2(H2O)4 + H2S
Hydrates can be produced by evaporation of an aqueous solution of zinc chloride. The temperature of the evaporation determines the hydrates For example, evaporation at room temperature produces the 1.33-hydrate.[17][21] Lower evaporation temperatures produce higher hydrates.[18]
Commercial samples of zinc chloride typically contain water and products from hydrolysis as impurities. Laboratory samples may be purified by recrystallization from hot dioxane. Anhydrous samples can be purified by sublimation in a stream of hydrogen chloride gas, followed by heating the sublimate to 400 °C in a stream of dry nitrogen gas.[22] A simple method relies on treating the zinc chloride with thionyl chloride.[23]
Reactions[edit]
The Zn2+2 ion[edit]
Molten anhydrous ZnCl2 at 500–700 °C dissolves zinc metal, and, on rapid cooling of the melt, a yellow diamagnetic glass is formed, which Raman studies indicate contains the Zn2+2 ion.[16]
Chloride complexes[edit]
A number of salts containing the tetrachlorozincate anion, [ZnCl4]2−, are known.[12] "Caulton's reagent", V2Cl3(thf)6] [Zn2Cl6], which is used in organic chemistry, is an example of a salt containing [Zn2Cl6]2−.[24][25] The compound Cs3ZnCl5 contains tetrahedral [ZnCl4]2− and Cl− anions,[7] so, the compound is not caesium pentachlorozincate, but caesium tetrachlorozincate chloride. No compounds containing the [ZnCl6]4− ion (hexachlorozincate ion) have been characterized.[7]
Adducts[edit]
The adduct with thf ZnCl2(thf)2 illustrates the tendency of zinc chloride to form 1:2 adducts with even weak Lewis bases. Being soluble in ethers and lacking acidic protons, this complex is used in the synthesis of organozinc compounds.[26]
The coordination complex ZnCl2(NH2OH)2 (zinc dichloride di(hydroxylamine)), known as Crismer's salt, releases hydroxylamine upon heating.[27]
When solutions of zinc chloride are treated with ammonia, various ammine complexes are produced. These include Zn(NH3)4Cl2·H2O and {chem2|ZnCl2(NH3)2}}.[28] The former contains the [Zn(NH3)6]2+ ion,[7] and the latter is molecular with a distorted tetrahedral geometry.[29] The species in aqueous solution have been investigated and show that [Zn(NH3)4]2+ is the main species present with [Zn(NH3)3Cl]+ also present at lower NH3:Zn ratio.[30]
Aqueous solutions of zinc chloride[edit]
Zinc chloride dissolves readily in water to give ZnClx(H2O)4−x species and some free chloride.[31][32][33] Aqueous solutions of ZnCl2 are acidic: a 6 M aqueous solution has a pH of 1.[16] The acidity of aqueous ZnCl2 solutions relative to solutions of other Zn2+ salts (say the sulfate) is due to the formation of the tetrahedral chloro aqua complexes such as [ZnCl3(H2O)]-.[34] Most metal dichlorides for octahedral complexes, with stronger O-H bonds. The combination of hydrochloric acid and ZnCl2 gives a reagent known as "Lucas reagent". Such reagents were once used a test for primary alcohols. Similar reactions are the basis of industrial routes from methanol and ethanol respectively to methyl chloride and ethyl chloride.[35]
In alkali solution, zinc chloride converts to various zinc hydroxychlorides. These include [Zn(OH)3Cl]2−, [Zn(OH)2Cl2]2−, [Zn(OH)Cl3]2−, and the insoluble Zn5(OH)8Cl2·H2O. The latter is the mineral simonkolleite.[36] When zinc chloride hydrates are heated, HCl gas evolves and hydroxychlorides result.[37]
Zinc oxychloride cement[edit]
Aqueous zinc chloride reacts with zinc oxide to form an amorphous cement that was first investigated in 1855 by Stanislas Sorel. Sorel later went on to investigate the related magnesium oxychloride cement, which bears his name.[38]
Decomposition[edit]
Anhydrous zinc chloride is able to melt and boil without any decomposition until 900 °C in an inert atmosphere. However, in the presence of oxygen, zinc chloride oxidizes to zinc oxide above 400 °C.[39]
When hydrated zinc chloride is heated, Zn(OH)Cl is produced instead of anhydrous zinc chloride:[40]
- ZnCl2·2H2O → Zn(OH)Cl + HCl + H2O
Cellulose dissolution in aqueous solutions of ZnCl2[edit]
Cellulose dissolves in aqueous solutions of ZnCl2. This unusual phenomenon is attributed to the formation of zinc-cellulose complexes, which have have been detected.[41] Cellulose also dissolves in molten ZnCl2 hydrate.[42] Overall, this behavior is consistent with Zn2+ as a hard Lewis acid]].
Conversion to other zinc compounds[edit]
Although zinc chlorides with diverse formulas and structuress exist, these salts often behave very similarly in aqueous solution. For example, solutions prepared from any of the hydrates of ZnCl2, as well as other halides (bromide, iodide), and the sulfate can often be used interchangeably for the preparation of other zinc compounds. These salts give precipitates o zinc carbonate when treated with aqueous carbonate sources:[5]
- ZnCl2 + Na2CO3 → ZnCO3 + 2 NaCl
Uses[edit]
Industrial organic chemistry[edit]
Zinc chloride is used as a catalyst or reagent in diverse reactions conducted on an industrial scale. Benzaldehyde, 20,000 tons of which is produced in Western countries, can be efficiently produced from inexpensive toluene by exploiting the catalytic properties of zinc dichloride. This process begins with the chlorination of toluene to give benzal chloride. A mixture of benzal chloride and anhydrous zinc chloride are treated continuously with water according to the following stoichiometry:[43]
- C6H5CHCl2 + H2O → C6H5CHO + 2 HCl
Similarly zinc chloride is employed in the main route to benzoyl chloride. It serves as a catalyst for the production of methylene-bis(dithiocarbamate).[5]
Organic syntheses[edit]
Zinc chloride is an occasional laboratory reagent often as a Lewis acid. A dramatic example is the conversion of methanol into hexamethylbenzene using zinc chloride as the solvent and catalyst:[44]
- 15 CH3OH → C6(CH3)6 + 3 CH4 + 15 H2O
This kind of reactivity has been investigated for the valorization of C1 precursors.[45]
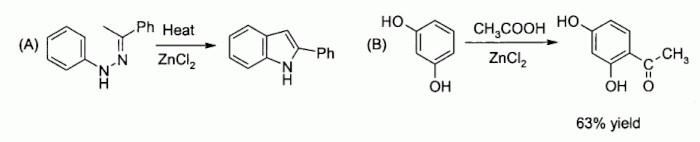
Examples of zinc chloride as a Lewis acid include the Fischer indole synthesis,[46] and Friedel-Crafts acylation reactions involving activated aromatic rings[47][48]
Related Lewis-acid behavior is illustrated by a traditional preparation of the dye fluorescein from phthalic anhydride and resorcinol, which involves a Friedel-Crafts acylation.[49] This transformation has in fact been accomplished using even the hydrated ZnCl2 sample shown in the picture above.

Zinc chloride also activates benzylic and allylic halides towards substitution by weak nucleophiles such as alkenes:[50]

In similar fashion, ZnCl2 promotes selective Na[BH3(CN)] reduction of tertiary, allylic or benzylic halides to the corresponding hydrocarbons.[22]
Zinc enolates, prepared from alkali metal enolates and ZnCl2, provide control of stereochemistry in aldol condensation reactions. This control is attributed to chelation at the zinc. In the example shown below, the threo product was favored over the erythro by a factor of 5:1 when ZnCl2.[51]

Organozinc precursor[edit]
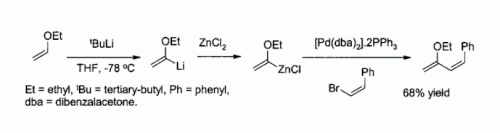
Being inexpensive and anhydrous, ZnCl2 is a widely used for the synthesis of many organozinc reagents, such as those used in the palladium catalyzed Negishi coupling with aryl halides or vinyl halides. The prominence of this reaction was highlighted by the award of the 2010 Nobel Prize in Chemistry to Ei-ichi Negishi.[52]
As a metallurgical flux[edit]
The use of zinc chloride as a flux, sometimes in a mixture with ammonium chloride (see also Zinc ammonium chloride), involves the production of HCl and its subsequent reaction with surface oxides.
Zinc chloride reacts with metal oxides (MO) to give derivatives of the idealized formula MZnOCl2.[53][additional citation(s) needed] This reaction is relevant to the utility of ZnCl2 solution as a flux for soldering — it dissolves passivating oxides, exposing the clean metal surface.[53] Fluxes with ZnCl2 as an active ingredient are sometimes called "tinner's fluid".[citation needed]
Zinc chloride forms two salts with ammonium chloride: [NH4]2[ZnCl4] and [NH4]3[ZnCl4]Cl, which decompose on heating liberating HCl, just as zinc chloride hydrate does. The action of zinc chloride/ammonium chloride fluxes, for example, in the hot-dip galvanizing process produces H2 gas and ammonia fumes.[54]
In textile and paper processing[edit]
Concentrated aqueous solutions of zinc chloride (more than 64% weight/weight zinc chloride in water) are capable of dissolving starch, silk, and cellulose.[citation needed]
Relevant to its affinity for these materials, ZnCl2 is used as a fireproofing agent and in fabric "refresheners" such as Febreze. Vulcanized fibre is made by soaking paper in concentrated zinc chloride.[citation needed]
Other uses[edit]
The zinc chloride smoke mixture ("HC") used in smoke grenades contains zinc oxide, hexachloroethane and granular aluminium powder, which, when ignited, react to form zinc chloride, carbon and aluminium oxide smoke, an effective smoke screen.[55]
Ninhydrin reacts with amino acids and amines to form a colored compound "Ruhemann's purple" (RP). Spraying with a zinc chloride solution forms a 1:1 complex RP:ZnCl(H2O)2, which is more readily detected as it fluoresces more intensely than RP.[56]
History[edit]
Zinc chloride has long been known but the industrial literature current applications evolved in the 20th century.
Dilute aqueous zinc chloride was used as a disinfectant under the name "Burnett's Disinfecting Fluid".[57] From 1839 Sir William Burnett promoted its use as a disinfectant as well as a wood preservative.[58] The Royal Navy conducted trials into its use as a disinfectant in the late 1840s, including during the cholera epidemic of 1849; and at the same time experiments were conducted into its preservative properties as applicable to the shipbuilding and railway industries. Burnett had some commercial success with his eponymous fluid. Following his death however, its use was largely superseded by that of carbolic acid and other proprietary products.[citation needed]
Natural occurrence[edit]
Anhydrous zinc chloride or its hydrates is not known in nature. However, the related zinc chloride hydroxide monohydrate is known as simonkolleite in nature.[59]
Safety and health[edit]
Zinc and chloride are essential for life. Zn2+ is a component of several enzymes, e.g., carboxypeptidase and carbonic anhydrase. Thus, aqueous solutions of zinc chlorides are rarely problematic as an acute poison.[5] Anhydrous zinc chloride is however an aggressive Lewis acid as it can burn skin and other tissues. Ingestion of zinc chloride, often from soldering flux, requires endoscopic monitoring.[60]
References[edit]
- ^ a b O'Neil, M. J.; et al. (2001). The Merck index : an encyclopedia of chemicals, drugs, and biologicals. N. J.: Whitehouse Station. ISBN 978-0-911910-13-1.
- ^ "Zinc chloride safety data sheet". Sigma-Aldrich. March 2, 2024. Retrieved May 21, 2024.
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0674". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Zinc chloride fume". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ a b c d e f Dieter M. M. Rohe; Hans Uwe Wolf (2007). "Zinc Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. pp. 1–6. doi:10.1002/14356007.a28_537. ISBN 978-3527306732.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 1211. ISBN 978-0-08-037941-8.
- ^ a b c d e Wells, A. F. (1984). Structural Inorganic Chemistry. Oxford: Clarendon Press. ISBN 978-0-19-855370-0.
- ^ Oswald, H. R.; Jaggi, H. (1960). "Zur Struktur der wasserfreien Zinkhalogenide I. Die wasserfreien Zinkchloride". Helvetica Chimica Acta. 43 (1): 72–77. doi:10.1002/hlca.19600430109.
- ^ Brynestad, J.; Yakel, H. L. (1978). "Preparation and Structure of Anhydrous Zinc Chloride". Inorganic Chemistry. 17 (5): 1376–1377. doi:10.1021/ic50183a059.
- ^ Brehler, B. (1961). "Kristallstrukturuntersuchungen an ZnCl2". Zeitschrift für Kristallographie. 115 (5–6): 373–402. Bibcode:1961ZK....115..373B. doi:10.1524/zkri.1961.115.5-6.373.
- ^ Mackenzie, J. D.; Murphy, W. K. (1960). "Structure of Glass-Forming Halides. II. Liquid Zinc Chloride". The Journal of Chemical Physics. 33 (2): 366–369. Bibcode:1960JChPh..33..366M. doi:10.1063/1.1731151.
- ^ a b Prince, R. H. (1994). King, R. B. (ed.). Encyclopedia of Inorganic Chemistry. John Wiley & Sons. ISBN 978-0-471-93620-6.
- ^ Ray, H. S. (2006). Introduction to Melts: Molten Salts, Slags and Glasses. Allied Publishers. ISBN 978-81-7764-875-1.
- ^ Danek, V. (2006). Physico-Chemical Analysis of Molten Electrolytes. Elsevier. ISBN 978-0-444-52116-3.
- ^ Price, D. L.; Saboungi, M.-L.; Susman, S.; Volin, K. J.; Wright, A. C. (1991). "Neutron Scattering Function of Vitreous and Molten Zinc Chloride". Journal of Physics: Condensed Matter. 3 (49): 9835–9842. Bibcode:1991JPCM....3.9835P. doi:10.1088/0953-8984/3/49/001. S2CID 250902741.
- ^ a b c Holleman, A. F.; Wiberg, E. (2001). Inorganic Chemistry. San Diego: Academic Press. ISBN 978-0-12-352651-9.
- ^ a b H. Follner; B. Brehler (1970). "Die Kristallstruktur des ZnCl2.4/3H2O" [The crystal structure of ZnCl2.4/3H2O]. Acta Crystallographica B (in German). 26 (11): 1679–1682. Bibcode:1970AcCrB..26.1679F. doi:10.1107/S0567740870004715.
- ^ a b E. Hennings; H. Schmidt; W. Voigt (2014). "Crystal structures of ZnCl2·2.5H2O, ZnCl2·3H2O and ZnCl2·4.5H2O". Acta Crystallographica E. 70 (12): 515–518. doi:10.1107/S1600536814024738. PMC 4257420. PMID 25552980.
- ^ Hamilton, R. T.; Butler, J. A. V. (1932). "Notes: The Preparation of Pure Zinc Chloride". Journal of the Chemical Society (Resumed): 2283-4. doi:10.1039/JR9320002282.
- ^ Goodwin, Frank E. (2017). "Zinc Compounds". Kirk-Othmer Encyclopedia of Chemical Technology. pp. 9–10. doi:10.1002/0471238961.2609140307151504.a02.pub3. ISBN 978-0-471-23896-6.
- ^ F. Mylius; R. Dietz (1905). "Über das Chlorzink. (Studien über die Löslichkeit der Salze XIV.)". Zeitschrift für anorganische Chemie. 44 (1): 209–220. doi:10.1002/zaac.19050440115.
- ^ a b Glenn J. McGarvey Jean-François Poisson Sylvain Taillemaud (2016). "Zinc chloride". Encyclopedia of Reagents for Organic Synthesis: 1–20. doi:10.1002/047084289X.rz007.pub3. ISBN 978-0-470-84289-8.
- ^ Pray, A. P. (1990). Anhydrous Metal Chlorides. Inorganic Syntheses. Vol. 28. pp. 321–322.
- ^ Mulzer, J.; Waldmann, H., eds. (1998). Organic Synthesis Highlights. Vol. 3. Wiley-VCH. ISBN 978-3-527-29500-5.
- ^ Bouma, R. J.; Teuben, J. H.; Beukema, W. R.; Bansemer, R. L.; Huffman, J. C.; Caulton, K. G. (1984). "Identification of the Zinc Reduction Product of VCl3 · 3THF as [V2Cl3(THF)6]2[Zn2Cl6]". Inorganic Chemistry. 23 (17): 2715–2718. doi:10.1021/ic00185a033.
- ^ Dashti, Anahita; Niediek, Katharina; Werner, Bert; Neumüller, Bernhard (1997). "Difluorenylzink als Alkylierungsmittel zur Darstellung von Triorganometallanen der 13. Gruppe. Synthese und Kristallstruktur von [GaFl3(THF)] · Toluol (Fl = Fluorenyl)". Zeitschrift für Anorganische und Allgemeine Chemie. 623 (1–6): 394–402. doi:10.1002/zaac.19976230163.
- ^ Walker, John E.; Howell, David M. (1967). "Dichlorobis(hydroxylamine)zinc(II) (Crismer's Salt)". Inorganic Syntheses. Vol. 9. pp. 2–3. doi:10.1002/9780470132401.ch2. ISBN 978-0-470-13240-1.
- ^ Vulte, H. T. (2007). Laboratory Manual of Inorganic Preparations. Read Books. ISBN 978-1-4086-0840-1.
- ^ Yamaguchi, T.; Lindqvist, O. (1981). "The Crystal Structure of Diamminedichlorozinc(II), ZnCl2(NH3)2. A New Refinement" (PDF). Acta Chemica Scandinavica A. 35 (9): 727–728. doi:10.3891/acta.chem.scand.35a-0727.
- ^ Yamaguchi, T.; Ohtaki, H. (1978). "X-Ray Diffraction Studies on the Structures of Tetraammine- and Triamminemonochlorozinc(II) Ions in Aqueous Solution". Bulletin of the Chemical Society of Japan. 51 (11): 3227–3231. doi:10.1246/bcsj.51.3227.
- ^ Irish, D. E.; McCarroll, B.; Young, T. F. (1963). "Raman Study of Zinc Chloride Solutions". The Journal of Chemical Physics. 39 (12): 3436–3444. Bibcode:1963JChPh..39.3436I. doi:10.1063/1.1734212.
- ^ Yamaguchi, T.; Hayashi, S.; Ohtaki, H. (1989). "X-Ray Diffraction and Raman Studies of Zinc(II) Chloride Hydrate Melts, ZnCl2 · R H2O (R = 1.8, 2.5, 3.0, 4.0, and 6.2)". The Journal of Physical Chemistry. 93 (6): 2620–2625. doi:10.1021/j100343a074.
- ^ Pye, C. C.; Corbeil, C. R.; Rudolph, W. W. (2006). "An ab initio Investigation of Zinc Chloro Complexes". Physical Chemistry Chemical Physics. 8 (46): 5428–5436. Bibcode:2006PCCP....8.5428P. doi:10.1039/b610084h. ISSN 1463-9076. PMID 17119651. S2CID 37521287.
- ^ Brown, I. D. (2006). The Chemical Bond in Inorganic Chemistry: The Bond Valence Model. Oxford University Press. ISBN 978-0-19-929881-5.
- ^ Kjonaas, R. A.; Riedford, B. A. (1991). "A Study of the Lucas Test". Journal of Chemical Education. 68 (8): 704. doi:10.1021/ed068p704.
- ^ Zhang, X. G. (1996). Corrosion and Electrochemistry of Zinc. Springer. ISBN 978-0-306-45334-2. Staff writer(s). "Simonkolleite Mineral Data". webmineral.com. Retrieved October 16, 2014.
- ^ Feigl, F.; Caldas, A. (1956). "Some Applications of Fusion Reactions with Zinc Chloride in Inorganic Spot Test Analysis". Microchimica Acta. 44 (7–8): 1310–1316. doi:10.1007/BF01257465. S2CID 96823985.
- ^ Wilson, A. D.; Nicholson, J. W. (1993). Acid-Base Cements: Their Biomedical and Industrial Applications. Cambridge University Press. ISBN 978-0-521-37222-0.
- ^ Frida Jones; Honghi Tran; Daniel Lindberg; Liming Zhao; Mikko Hupa (2013). "Thermal Stability of Zinc Compounds". Energy & Fuels. 27 (10): 5663–5669. doi:10.1021/ef400505u.
- ^ House, J. E. (2008). Inorganic Chemistry. Academic Press. ISBN 978-0-12-356786-4.
- ^ Xu, Q.; Chen, L.-F. (1999). "Ultraviolet Spectra and Structure of Zinc-Cellulose Complexes in Zinc Chloride Solution". Journal of Applied Polymer Science. 71 (9): 1441–1446. doi:10.1002/(SICI)1097-4628(19990228)71:9<1441::AID-APP8>3.0.CO;2-G.
- ^ Fischer, S.; Leipner, H.; Thümmler, K.; Brendler, E.; Peters, J. (2003). "Inorganic Molten Salts as Solvents for Cellulose". Cellulose. 10 (3): 227–236. doi:10.1023/A:1025128028462. S2CID 92194004.
- ^ . doi:10.1002/14356007.a03_463.pub2.
{{cite journal}}: Cite journal requires|journal=(help); Missing or empty|title=(help) - ^ Chang, Clarence D. (1983). "Hydrocarbons from Methanol". Catal. Rev. - Sci. Eng. 25 (1): 1–118. doi:10.1080/01614948308078874.
- ^ Olah, George A.; Doggweiler, Hans; Felberg, Jeff D.; Frohlich, Stephan; Grdina, Mary Jo; Karpeles, Richard; Keumi, Takashi; Inaba, Shin-ichi; Ip, Wai M.; Lammertsma, Koop; Salem, George; Tabor, Derrick (1984). "Onium Ylide chemistry. 1. Bifunctional acid-base-catalyzed conversion of heterosubstituted methanes into ethylene and derived hydrocarbons. The onium ylide mechanism of the C1 → C2 conversion". J. Am. Chem. Soc. 106 (7): 2143–2149. doi:10.1021/ja00319a039.
- ^ Shriner, R. L.; Ashley, W. C.; Welch, E. (1942). "2-Phenylindole". Organic Syntheses. 22: 98. doi:10.15227/orgsyn.022.00981955; Collected Volumes, vol. 3, p. 725.
- ^ Cooper, S. R. (1941). "Resacetophenone". Organic Syntheses. 21: 103. doi:10.15227/orgsyn.021.0103; Collected Volumes, vol. 3, p. 761.
- ^ Dike, S. Y.; Merchant, J. R.; Sapre, N. Y. (1991). "A New and Efficient General Method for the Synthesis of 2-Spirobenzopyrans: First Synthesis of Cyclic Analogues of Precocene I and Related Compounds". Tetrahedron. 47 (26): 4775–4786. doi:10.1016/S0040-4020(01)86481-4.
- ^ Furnell, B. S. (1989). Vogel's Textbook of Practical Organic Chemistry (5th ed.). New York: Longman/Wiley.
- ^ Bauml, E.; Tschemschlok, K.; Pock, R.; Mayr, H. (1988). "Synthesis of γ-Lactones from Alkenes Employing p-Methoxybenzyl Chloride as +CH2-CO2− Equivalent" (PDF). Tetrahedron Letters. 29 (52): 6925–6926. doi:10.1016/S0040-4039(00)88476-2.
- ^ House, H. O.; Crumrine, D. S.; Teranishi, A. Y.; Olmstead, H. D. (1973). "Chemistry of Carbanions. XXIII. Use of Metal Complexes to Control the Aldol Condensation". Journal of the American Chemical Society. 95 (10): 3310–3324. doi:10.1021/ja00791a039.
- ^ Negishi, Ei-Ichi (2011). "Magical Power of Transition Metals: Past, Present, and Future (Nobel Lecture)". Angewandte Chemie International Edition. 50 (30): 6738–6764. doi:10.1002/anie.201101380. PMID 21717531.
- ^ a b Wiberg, Nils (2007). Lehrbuch der Anorganischen Chemie [Holleman & Wiberg, Textbook of Inorganic chemistry] (in German). de Gruyter, Berlin. p. 1491. ISBN 978-3-11-017770-1.
- ^ American Society for Metals (1990). ASM handbook. ASM International. ISBN 978-0-87170-021-6.
- ^ Sample, B. E. (1997). Methods for Field Studies of Effects of Military Smokes, Obscurants, and Riot-control Agents on Threatened and Endangered Species. DIANE Publishing. ISBN 978-1-4289-1233-5.
- ^ Menzel, E. R. (1999). Fingerprint Detection with Lasers. CRC Press. ISBN 978-0-8247-1974-6.
- ^ Watts, H. (1869). A Dictionary of Chemistry and the Allied Branches of Other Sciences. Longmans, Green.
- ^ McLean, David (April 2010). "Protecting wood and killing germs: 'Burnett's Liquid' and the origins of the preservative and disinfectant industries in early Victorian Britain". Business History. 52 (2): 285–305. doi:10.1080/00076791003610691. S2CID 154790730.
- ^ "Simonkolleite". mindat.org. Retrieved 20 February 2024.
- ^ Hoffman, Robert S.; Burns, Michele M.; Gosselin, Sophie (2020). "Ingestion of Caustic Substances". New England Journal of Medicine. 382 (18): 1739–1748. doi:10.1056/nejmra1810769. PMID 32348645.
Further reading[edit]
- N. N. Greenwood, A. Earnshaw, Chemistry of the Elements, 2nd ed., Butterworth-Heinemann, Oxford, UK, 1997.
- Lide, D. R., ed. (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- The Merck Index, 7th edition, Merck & Co, Rahway, New Jersey, USA, 1960.
- D. Nicholls, Complexes and First-Row Transition Elements, Macmillan Press, London, 1973.
- J. March, Advanced Organic Chemistry, 4th ed., p. 723, Wiley, New York, 1992.
- G. J. McGarvey, in Handbook of Reagents for Organic Synthesis, Volume 1: Reagents, Auxiliaries and Catalysts for C-C Bond Formation, (R. M. Coates, S. E. Denmark, eds.), pp. 220–3, Wiley, New York, 1999.